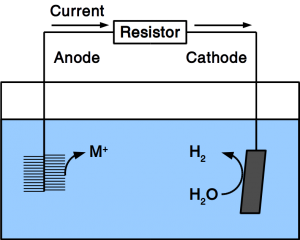
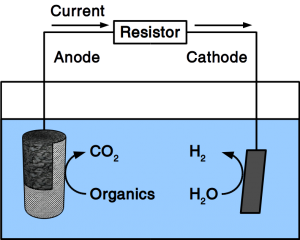
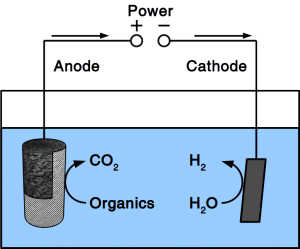
As our preferred reader, you know already that we work on Power-to-Gas to combat Global Warming. We think that giving CO2 a value will incentivize its recycling and recycling it into fuel turns it into a commodity that everyone needs. While the price of CO2 from air is still too high to convert it into combustion fuel, working on the other end (the CO2 conversion) will help to accommodate such high prices. We have now published an research paper that shows how how to reduce the costs of electronic equipment needed for CO2 conversion. For Power-to-Gas as well es for electrosynthesis of liquid fuels, it is necessary to poise an electrochemical potential. So far, only electronic potentiostats could do that. We have developed a software solution that can control cheap off-the-shelf hardware to accomplish the same goal. Since the software controls µA as well as MA, it is freely scalable. By stacking cheap power supplies, it can also run unlimited channels.

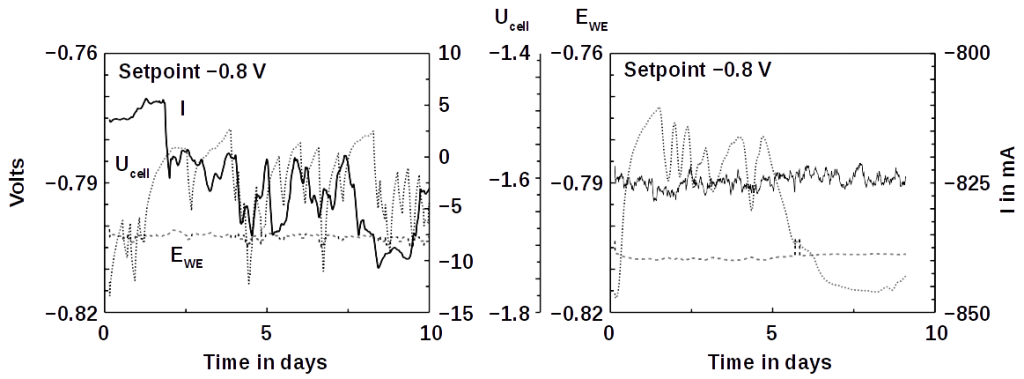
We tested the software at a typical experimental Power-to-Gas setup at −800 mV and found that the recorded potential was stable over 10 days. The small electrochemical cells could also be replaced by a larger 7 liter reactor treating real wastewater. The potential was stable as well.
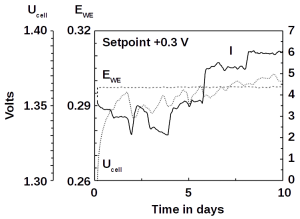
As instrument control of mass products also makes the chemical processes involved cheap, microbial electrolysis of wastewater becomes economically feasible. Removal of wastewater organics usually occurs at positive electrochemical potentials. Indeed, the software also stabilizes such potentials at +300 mV.
The potentiostat is currently available as command line version. We are currently accepting pre-orders at a 50% discount for the commercial version that comes with a graphical user interface and remote control using an internet browser.