
How do we shape human exploration on Mars to minimize what we must bring with us and to maximize the value and utility of what we bring, and augment it with what is already there?
To provide sufficient fuel for a safe return of the Mars crew, we can produce methane and oxygen on Mars which could be aided by microbes that are brought to Mars. We suggest lightweight perovskite solar panels that may be transported to Mars. In the optimistic scenario, about 18 months are required for the installation of surface solar power and fuel production for the failsafe return after which the crewed mission commences. The pessimistic scenario takes 4 years. To save oxygen, we also suggest Martian perchlorates as rocket fuel component. For later missions’ food supply, we suggest to use lichens as primary colonizers to produce organic rich soil.
Procedure to produce energy on Mars
We suggest the use of in situ Martian resources for the production of methane as ascent and return propellant. Since oxygen generated during electrolysis is not sufficient for a safe return, we also suggest to use algae for the production of oxygen. Algal biomass will be used as soil for food production. Methane producing microbes will be contained in methanogenic electrolysis reactors (MER) while algae will grow in covered craters. The production of methane on Mars is to be carried out autonomously by robots and reactors that will land near the ice-rich polar regions to melt water as electrolyte for low temperature electrolysis. The Mars lander will autonomously construct facilities with the purpose of propellant production to fuel the Mars transfer vehicle which enables transport between Mars surface and Earth orbit. Only when enough propellant for a safe return to Earth has been produced, shall a manned Mars mission begin. Furthermore, methane will be used as energy storage should there be failure of energy collectors such as solar panels. Goal is to erect a 3.5 MW solar power plant on Mars by the end of the first manned mission.
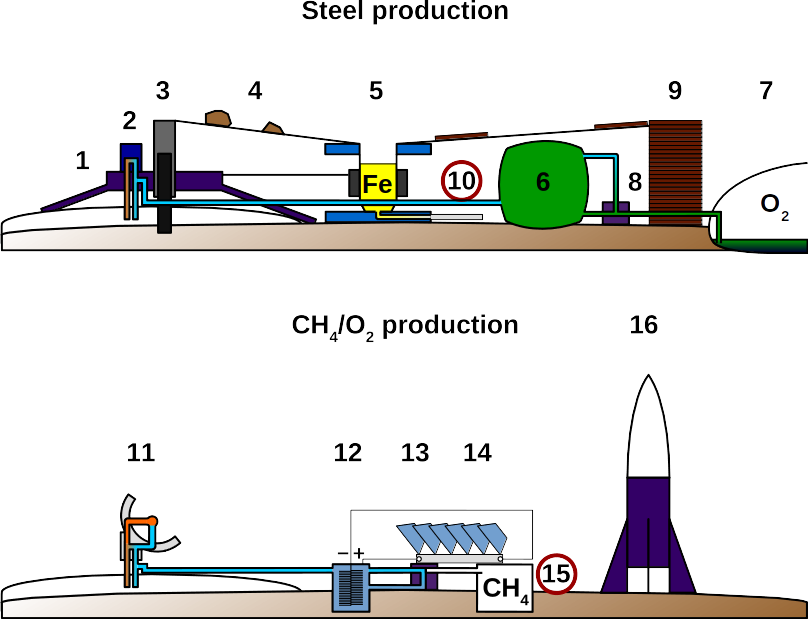
To sustain the life of 6 crew members a power production capacity of 170 kW (see “Surface habitat energy needs”) is required and has highest priority along with fuel production for a safe journey home. The procedure is sketched out in the figure above. It is a stepwise process during which most steps are interdependent and therefore self-accelerating as power production increases. To minimize risk of failure, we recommend at least four independent landers in the circum-polar region of Mars. The polar regions bear the highest surface water content which is crucial for manned explorer missions, methanogenic electrolysis as well as the production of oxygen and biomass. The landers will carry a small nuclear fission device that will begin mining for iron and titanium ores to produce the steel required as structural support for solar panels. First, the lander begins mining for iron ores so that steel production can begin. Graphite or other reduced forms of carbon for steel production will be shipped from Earth because organic matter production on Mars by algae is a slow process. Alternatively, lightweight modular carbon fiber elements to mount solar panels will be brought from Earth to Mars. Once the 170 kW solar plant is established, melting ice for the methanogenic electrolysis reactors (MER) begins. Combined power from the nuclear fission reactor (NFR) and the solar plant will be used. Only when the amount of methane for a failsafe return (Orion capsule size) has been produced, power will be diverted into three equal parts: continue to melt ice for algae, start or continue to mine ores for thermal collectors, continue to produce methane. After enough thermal collectors are produced to pace ice melting with algal growth, electrical ice melting will be shut off. Now, electrical energy is used for steel production to install more solar panels and methane production until sufficient for a comfortable (more payload) return. By this time, the crew is arriving and decides what the highest priorities are. We recommend to focus on accelerating algal growth for oxygen and biomass production as complete independence from Earth requires the production of organic carbon from CO2.
Energy need for the Mars Transfer Vehicle
Fuel produced on Mars will serve 3 purposes:
- Failsafe: return of one Orion-sized vehicle
- Desired: production of return fuel allowing for less stress (more comfort) during transit
- Energy storage at night or power failure
Two options for Mars-Earth return seem possible. (1) Option one was suggested by NASA’s DRA 5.0 and involves one Orion-like vehicle of about 12 tons and a travel speed of 14 km/s. This option requires only one vehicle but provides less comfort for the long journey home and hence, it exposes the crew to higher stress. However, it uses less fuel therefore enables a faster completion of the first Mars mission. (2) We envision a second option that involves two vehicles, one Orion-like lift-off vehicle for transport of 6 crew members into a Mars orbit of 250 km and one transit vehicle to return to Earth. As option two is the preferred option, we recommend to use option one, the Orion-only scenario, as failsafe scenario.
We assume the capacity of a manned Orion capsule with Dragon thrusters (Draco) as reference. The Orion-sized vehicle can carry 6 crew members and has a weight of 12 tons including fuel. For a Mars lift-off, a thrust-mass ratio of at least 5 N/kg will be needed resulting in 60 kN thrust or 150 Draco thrusters to lift 6 crew members. The travel time from Martian surface to a 250 km orbit would be 7 minutes at full thrust. About 600 m3 methane (at Earth atmospheric pressure) would be required for the lift-off. To produce this amount of methane at 210 kW (40 kW nuclear fission and 170 kW solar power, see “Surface habitat energy needs”), 3 years of dedicated fuel production are necessary. The proposed solar power installations of 1,400 m2 perovskite solar cells can produce effectively 170 kW during a Mars day of 8h (that is, 20 kW/m2 can be captured by perovskite panels). Using the same Orion capsule also for the Mars-Earth transit, another 7 minutes or 17,000 km are required to reach the travel speed of 14 km/s and approximately the same for full stop. To produce enough fuel for the Mars-Earth transit, only 3 more days are required. Once enough fuel for the failsafe scenario has been produced, the crew will leave the lower Earth orbit heading for Mars.
For the comfort scenario, we assume the proposed 63-ton crewed payload option from the Mars DRA 5.0 used for Earth-Mars transit. This seems reasonable as most equipment will be left behind on Mars and only the transit habitat, the Orion capsule (for emergencies), engines and fuel are needed. This scenario, however, requires considerably more fuel for which the 210 kW surface power generators must produce methane for at least 42 years. Since this is out of scope, we recommend an extension of the surface power production to 3,500 kW which would reduce the required production of methane to 30 months at 14 km/s travel speed or 12 months at 9 km/s. The reduced stress on the crew justifies the lower travel speed and the higher investment. However, 6.3 tons (corresponding to 0.18 km2) of perovskite solar panels will be required to produce sufficient fuel. About 280 tons of steel are necessary as structural support for this scenario. Since 900 kWh/ton of steel will be consumed for electric smelting, only about one year of steel production using a 40 kW NFR (or 2 months using the full combined 210 kW) justify the increased comfort. The shipment of carbon fiber elements form Earth may completely eliminate the need for steel production in this stage of the mission. The process will be self-accelerating as power production increases during the solar panel assembly process.
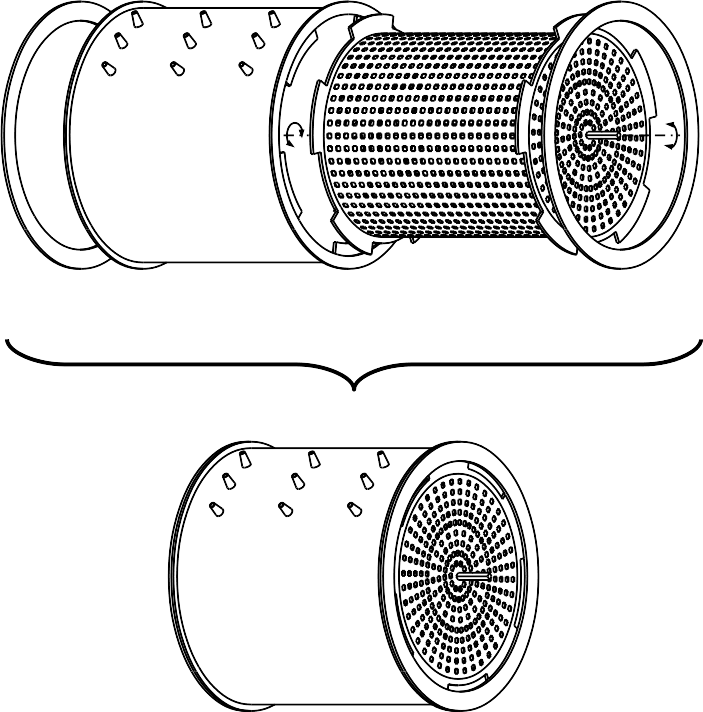
To produce methane sufficient for one lift-off, a 200,000 liter MER is required using steel mesh/brush electrodes (anode and cathode) of 2,200 m2 projected surface (see figure below). A modular redundancy system of smaller dimension will improve safety but requires more material. MERs have the theoretical capacity to produce sufficient ascent fuel for one Orion capsule within less than one or two days, if power supply were sufficient. At maximum performance, this reactor would consume about 100 GWh or 220 kWh/mol methane. Microbes will facilitate electrolysis at low temperatures and these microbes will be carried by the Mars lander in small (100 mL) redundant batches. Since the limit for methane production is not the reactor capacity but the electrical power available, doubling the amount of solar panels will half the time required for methane production and so on. To store the so produced methane we recommend to first pressurize water to 200 bar before it is injected into the MER. To extract as from the electrolyte, a small pressure reduction is needed and the so obtained gas phase is then conducted into pressurized steel tanks for later use.
An experimental MER needs to be constructed on Earth to prove this concept. Like the Mars reactor, this experimental MER will be a 5 x 5 m cylindrical reactor of either one or two chambers each. The advantage of the two-chamber system is the separation of oxygen and methane but it requires more water while the single chamber reactor is easier to build and holds less water but O2/CH4 separation is required after production. Unfortunately, the ratio of oxygen to methane is difficult to predict as it depends on the anodic pH. A mass ration greater than 2:1 is required. We therefore propose the use of algae as additional oxygen source (see “Photosynthesis crater to produce oxygen and biomass”). As electrodes brush or spiral steel mesh electrodes will be used. Steel mesh (40 x 40 mesh) produced on Mars will be used having a 1,100 m2 projected surface of each electrode.
Alternative oxidants in cold methane fuel cells or rocket fuel
It is anticipated that oxygen scarcity imposes severe limitations on any manned Mars mission. Oxygen is crucial as propellant and for any human presence. The use of methane for energy storage makes only sense if there is an adequate electron acceptor. While methane can be burnt in turbines at acceptable efficiencies for electricity production, it may also be used in fuel cells. However, no catalysts exist that oxidize methane on electrodes at room temperature or below. The only possible exception are anaerobic methane oxidizing consortia that naturally use biological electron transport chains. The use of biological electron transport chains opens the possibility to capture the energy stored in electrons during transport. Since this is electron acceptor independent, oxidized metal minerals, which are abundant on Mars, can be used as electron acceptors. The disadvantage of this methane fuel cells is that less energy will be captured compared with oxygen. Also, they only exist in theory.
(1) CH4 + 2 O2 → HCO3− + H+ + H2O ;∆G°’ = −830 kJ/molCH4
(2) CH4 + 4 Fe2O3 + 15 H+ → HCO3− + 8 Fe2+ + 9 H2O ;∆G°’ = −250 kJ/molCH4
The high acidity on Mars, however, is in favor of reaction, forming additional water from iron oxides and protons using the reductive power of methane. Soluble Fe2+ may be used for electric steel production as the reduction of Fe2+ to Fe0 requires a considerably lower redox potential and therefor lower energy.
Martian perchlorate salts may serve as oxidant in rocket fuel. Ammonium perchlorate and, on Mars, the more prevalent calcium perchlorate are explosive oxidizers. To transform calcium perchlorate into the ammonium salt, ammonium can be produced by a variety of microbial process such as nitrogen fixation (by way of the nitrogenase enzymes) and catabolic ammonification of amino acids or waste urea (by the urease enzyme). Should collection and compression of the photosynthetically derived O2 gas prove impractical for in rocket fuel, our solid oxidizer approach is also submitted. This dual oxidizer strategy will provide for far greater flexibility and more breathable oxygen. Mined perchlorate may also be used to disinfect water.
Photosynthesis crater to produce oxygen and biomass
Oxygenic biophotolysis of water using psychrophilic (cold-loving), dinitrogen fixing cyanobacteria, i.e. blue green algae, grown in covered craters is one proposed plausible means of generating the need for oxygen and biomass. The surplus of oxygen will be required as propellant and component of artificial air in the surface habitat (SHAB). While initially not crucial for a Mars mission, the production of organic matter is useful for more extended missions with larger teams and longer presence. Organic matter is essential for rich soil which, in turn, is pivotal for vegetable food production on Mars. Moreover, cyanobacteria and algae require little engineering and energy which makes them ideal for autonomous production of utility compounds such as organic matter and oxygen.
The amount of damaging cosmic rays and UV can be higher due to the lack of an ozone layer and protective magnetosphere. The amount of cosmic radiation (est. 0.076 Grays per year) is certainly within the tolerable range for many Earthly microbes as it is only around what the interior of the international space station is exposed to. UV light, with its shorter wavelength, can be readily blocked by a thin covering of Martian soil whereas longer wavelengths of photosynthetically active radiation can penetrate further. The microbes will be selectively enriched in their survival zones. Alternatively, a UV protective cover could be used over the crater. The lightweight but durable and robust crater coverings could take the form of an inflatable inverted dome anchored around the crater edge by cables and spikes. The clear upper canopy would admit sunlight but have coating to block harmful radiation while the curved lower surface could be reflective (to maximize photosynthesis) or black to absorb heat. Solar powered gas pumps could adjustably increase the internal gas pressure to accelerate carbon and nitrogen fixation rates and water accumulation from the trace water vapor available.
Conversion of limited amount of solar energy and frozen water plus copious CO2 into biologically generated oxygen plus organic matter will require a phototroph capable of survival at extremely low temperatures consistent with the Martian surface. We propose to identify terrestrial cyanobacteria capable of this by selectively enriching them from mixed biofilm consortia obtained from the Earth’s Arctic and Antarctic regions. Samples obtained from rocky coastal brines will be subjected to intensive evaluation in selective enrichment freezers outfitted to replicate the polar Martian habitat. The finding that the lichen Pleopsidium chlorophanum (gold cobblestone lichen) can survive, adapt and grow under Martian environmental conditions bodes well for this approach.
During the initial surface resource utilization phase (see figure above), the growth of algae is the most time consuming step and therefore the production of liquid water has highest priority. The use of craters will eliminate the need for containers for growing and there reduce the amount of material brought to Mars. Ideally, such craters are equatorial flat water ponds that ensure maximum sunlight capture and minimum water reheating. These preconditions do not align with the initial mission setup (landing near polar ice caps) but should be prepared during the first mission. That is, water pipes from the circum polar regions to the equatorial areas must be constructed. The pipes may need to be heated which requires additional energy or heating the melted water to high temperatures and pressures to prevent ice formation during transport.
Production of water as medium for methanogenic electrolysis and algae
The lack of liquid water is a major hindrance since active metabolism requires a fluid aqueous medium. In addition to the production of methane fuel, melting ice is the greatest challenge for the first manned Mars mission. Liquid water is essential for MERs and algae craters. Hence, all excess of heat or electrical power produced should be directed to melting of ice after methane fuel production is secured. The obtained CO2-rich brine is the electrolyte for MERs. The high acidity is not inhibitory for microbial growth as acidophilic methanogens and algae were reported from terrestrial environments. Fortunately, the low pH will reduce the electrical overpotential needed for hydrogen generation, which is the intermediate step during methanogenic electrolysis. The low pH, on the other hand, inhibits oxygen formation which is why corrosion of steel anodes is anticipated to become a possible problem. Anode corrosion must be monitored and shall not exceed a certain, yet to be determined, threshold before spent anodes are recycled in steel foundries brought with the first mission.
The low temperatures on Mars that reach only 20°C in equatorial regions also impose a major hurdle on liquid water maintainance. That is, water may need to be heated by parabolic heat collectors to remain liquid. However, Fischer et al. recently found that “when the salts are in contact with water ice, liquid brine forms in minutes, indicating that aqueous solutions could form temporarily where salts and ice coexist on the Martian surface and in the shallow subsurface.” If our crater canopy is fitted with an internally reflective coating in the infrared spectrum small green houses can be created an the brine will remain liquid longer.
The MERs use methanogenic microorganisms for methane production which will be brought to Mars along with algae seeds by the landers. The methanogenic microbes are highly efficient in methane production, resulting in electricity capturing efficiencies close to 100%. Precious metal catalysts are not required. In contrast, for effective oxygen production, platinum or palladium coating may be required on the anodic side of the MERs. Anodic algae appear to be an alternative but need to be further explored. Since the amount of platinum used is very low, it may be transported as salt to Mars and electroplated on steel electrodes once they are ready. Electroplating is an easy procedure so that a robot can accomplish this task within few minutes. However, platinum recycling requires 1-2 days of work of one crew member.
About 280 tons of steel for structural support of solar panels are required (see “Production of steel for structural support of Mars surface components”). The carbon content of steel should not exceed 2.1% to guarantee high stability and therefore we chose 1.5% carbon for Mars steel. That is, about 4 metric tons of carbon are required for steel production. This is the bottleneck of steel production. Assuming cold conditions on Mars comparable to the Antarctic, a good approximation for biomass concentration in brine is 5 mg/m3. At this concentration, nearly 1 billion m3 water need to be processed. While the existing amount of 821,000 km3 would be more than sufficient, it is impossible to melt this amount of ice within the mission’s timeframe using an NFR of 40 kW even if other power sources were counted in. Therefore, parabolic heat collectors could be shipped from Earth as well. At an energetic efficiency of parabolic heat collectors of 80%, 300 tons would be required to melt this amount water within 2 years. Using 10 tons of parabolic collectors, one can melt only 26,000 m3 for algae during 2 years. This is enough to produce 130 g algal carbon in little more than 2 years assuming a constant concentration of 5 mg/m3. It is more efficient to bring 4 tons of graphite to Mars for initial steel production or consider reflecting surfaces other than polished steel.
Alternative use of covered craters to accumulate water using native perchlorates
In light of the considerable difficulties associated with installing long water pipelines to fill craters with water, we outline an elegant alternative strategy for gradually capturing water from the atmosphere using native perchlorates in the Martian sediments.
Perchlorate salts have been detected in Martian sediments and craters such as the Dale Crate and at concentrations of 0.5-1% globally. Calcium perchlorate is an extremely hygroscopic component of the Martian soil that was recently discovered to cyclically draw H2O from the Martian atmosphere into the soil by night to form saline liquid brine. By sealing the crater covers by day, when the water normally sublimates off, and then opening in-current or one way valves by night after photosynthetically formed O2 has been recovered, atmospheric water vapor can slowly be accumulated as brine liquid/ice inside of the crater at zero or minimal energetic cost.
Halophilic algae tolerate high salt concentrations and low temperatures. Due to the high concentration of CO2 in the native atmosphere the crater covering will amplify the warming effects of this greenhouse gas to prolong the duration of liquid state water needed for nitrogen and carbon fixation. By transporting more soil-derived perchlorates, possibly with crushed water ice deposits, into the covered crater, water can slowly be accumulated. Biologically formed nitrous oxide gas might further accentuate the internal greenhouse warming and thus biological rates of activity.
Perchlorates salts from inside the crater can be recovered from the liquid brine to gradually reduce the salinity of the water. This could be done using parabolic evaporation troughs periodically lifted above the briny surface. Since perchlorates are considered a human toxin, they can be removed by some microbes such as perchlorate reducing bacteria (PRBs) which use percholorates as alternative electron acceptors. Such PRBs could be introduced at a later stage to eventually render the water-filled covered craters non toxic to higher forms of life.
Soil conditioning through phototrophic primary productivity
Lichen and blue green algae have both been used as foods on Earth for hundreds of years. Spirulina is one example of a widely consumed cyanobacterium that uses sunlight to synthesize essential vitamins, antioxidants like beta-carotene and fatty acids from CO2. One major advantage of using a nitrogen fixing cyanobacterium is that they can use solar energy to convert atmospheric nitrogen gas directly into the essential amino acids that future manned missions will need to build and maintain muscle on the Red planet. This will reduce the amount of fuel spent on shuttling food supplies in. Surprisingly, some species of cyanobacteria contain 60% protein per dry gram which is more protein than beefsteak, without the high amount of deleterious cholesterol. Gaseous nitrogen makes up roughly 2.7% of Mars’ thin atmosphere and is available globally. Nitrogen gas is not the only bioavailable form of nitrogen needed to grow oxygenic phototrophs. Nitrates are an ideal fertilizer. The Curiosity rover identified bioavailable nitrates as a significant component of the sediment on Mars. Trace elements are also present in rocks and soil but may require processing.
Soil conditioning of the Martian landscape initiated by this pioneer mission would be required for subsequent longer term human habitation and colonization. Lichen and cyanobacteria are common pioneer species on Earth that grow in the rocky wake of retreating glaciers. These phototrophs are known to accelerate rock weathering and to facilitate the release of essential minerals. Phosphorous, much like nitrogen, is an important macro-phytonutrient that is now known to be a significant component of the Martian surface. Indeed, some nitrogen fixing cyanobacteria can up-regulate their expression of phosphorous liberating phytase enzymes when exposed to phosphorus limitation. Cyanobacteria also build and stabilize soils by reducing their susceptibility to wind erosion through formation of organic extracellular polysaccharides that help trap and retain moisture. Lichen can also release acids and metabolites that contribute to rock break down and soil formation. While lichens and cyanobacteria may adapt to higher UV light dosages on Mars, they can be protected initially by a thin cover as described in “Photosynthesis crater to produce oxygen and biomass”.
Production of steel for structural support of Mars surface components
Steel cannot be brought to Mars as there will be at least 2.2 tons of steel necessary for structural support for 1,400 m2 perovskite solar panels. While lightweight carbon fiber modules could be used as structural support, it is possible to produce steel in situ. Steel production on Mars seems an apparent alternative to transport of construction material given the abundance of iron, nickel and titanium on Mars. However, it also requires organic carbon which is to be produced by CO2-fixing algae that grow first in enrichment tanks (transparent plastic bags) and later in covered craters. After dehydrating the algae medium, recycling and reheating it, dry algae pellets will be used as supplement for steel production. The dehydration and reheating process require additional energy which can be provided as heat using parabolic collectors. Parabolic collectors are more efficient in terms of energy capturing and easier to construct as polished steel can be used as opposed to organic Pb/I composites in perovskite solar cells. Steel is then shaped and polished to build parabolic thermal collectors to melt more ice and provide more energy until the NFR and solar panels can be fully replaced by parabolic collectors that can also produce electricity. The production of steel is limited by the amount of organic carbon available. Therefore, we recommend to explore the possibility to use methane gas as reductant and carbon source for steel production. Methane gas production is faster and requires less water resources than algae.
The Mars landers will also mine iron ores and silicates for the production of wires, solar panels and construction materials. Steel will be produced in an induction furnace using iron ores and graphite or organic biomass. Organic biomass from algae tanks will be used for steel production. This organic biomass will also be used for graphite production at a later stage of the mission. Alternative furnace concepts are possible. For example, methane can be used as reductant. Another alternative would be an electric arc furnace or sacrificial graphite electrodes. Graphite can be produced from organic carbon as follows
- Organic carbon from CO2 by cold adapted algae
- Organic carbon + 800ºC → C
- C + SiO2 + 1,400ºC → SiC
- SiC + 4,200ºC → graphite
The steel and graphite induction furnaces will be carried by the landers
Energy for initial steel production for the construction of the 170 kW solar plant (see “SHAB energy needs”) is produced by an NFR. A 40 kW reference NFR is recommended. Steel production from iron ore using electric smelting requires 900 kWh per ton of steel. That is, to produce enough structural support for solar panels for 6 people, about 2,000 kWh are necessary or about 3 days of energy production at full performance. This is based on the assumption that steel of 2 mm thickness and 10% of the perovskite area of 1,400 m2 is sufficient. To produce enough structural support for 3.5 MW (0.18 km2) perovskite solar cells needed for the comfortable return option, 280 tons of steel are required. At 50 days of steel production using the entire 210 kW (40 kW NFR + 170 kW solar perovskite life support) are anticipated. To add 1.5% carbon, 4 tons of graphite are necessary which will be carried as cargo from Earth.
Steel is necessary for parabolic heat collectors on site as well. Parabolic heat collectors are required for melting ice for algae growth. The production of steel sufficient for parabolic collectors to melt 1 billion m3 of ice is approximately 600 tons, i.e. 9 additional tons of graphite need to be shipped. To make this amount of steel on Mars another 2 years will be necessary at least. This appears to be the best trade-off between cargo transport and waiting time for a Mars mission but is still an approximation. The algal side product oxygen, also justifies this approach. Since the launch of a manned mission is not dependent on algae production, it is not counted as wait time, which would add another 4 years prior to launch. This shall only demonstrate the feasibility of in situ steel production on Mars. As alternative, the use of methane produced on Mars as carbon and electron source for steel needs to be explored as this may eliminate the need for graphite transport or carbon fiber construction materials are transported to Mars.
Possible perovskite production and reuse of lead produced by the nuclear fission reactor
Lead remnants from the 235U nuclear fission reactor aboard the landers can be used as they are a side product of the radioactive decay of contaminating 238U. There are no confirmed higher concentrations of iodine on Mars and this element needs to be brought in the form of elemental iodine, KI or NaI with the lander to produce the PbI and methyl ammonium iodide. However, since iodine can be replaced by the element chlorine for perovskite production, the initial amount iodine may not need to be replenished because chlorine is an abundant element on Mars.
Solvents required for perovskite cells can be produced in situ using methane gas and acetic acid (also a possible side product of MERs) as precursors as soon as they become available. To bridge the intermediate gap, solvents and organic reactants brought to Mars by the landers may be used. These materials are:
- N,N-dimethylformamide (solvent)
- 2-propanol (solvent)
- <ethylammonium iodide (reactant)
- 2,2′,7,7′-tetrakis(N, N-di-p-methoxyphenylamine)-9,9′-spirobifluorene (spiro-MeOTAD, reactant)
As synthesis of perovskite cells may still be to difficult on Mars, we recommend the transport of these components. As shown below (Surface habitat [SHAB] energy needs), only 9 kg perovskite solar panels will be required. As photo voltaic systems are constantly improved, one can expect better performance and lighter weights in future.
Surface habitat (SHAB) energy needs
The average energy need per capita in the European Union was 150 GJ/year. Although this estimate is likely too high for a permanent colony on Mars we used this as reference herein. A perovskite solar panel operating at 12% efficiency can produce 14 MJ/day/m2 from Mars aphelion 8 hour solar radiation assuming 170 W/m2. That is, 240 m2 methyl ammonium lead halide perovskite solar panels are required to sustain the presence of one person on Mars. This requires 350 g mesoporous TiO2, 370 g Au per capita. The light weight of about 720 g per capita makes a transport of these components from Earth to Mars feasible (8.5 kg total). Mounting the thin solar power collector on a stable steel surface can be achieved on Mars but only if steel is produced in situ. NASA’s Human Exploration of Mars Design Reference Architecture recommends a mission of 6 explorers. That is, at about 1,400 m2 perovskite solar panels or 170 kW capacity will be required only to sustain life of one manned explorer mission. Before installation of these panels, steel will have to be produced on which they can be mounted. For the envisioned larger energy needs, 3.5 MW are necessary and for this, about 6.3 tons of perovskite may be shipped to Mars.
Compounds produced on Mars (purpose in brackets)
- Iron, Fe0 (steel)
- Steel (construction, wires, electrodes)
- Graphite (steel, electrodes)
- Silicium dioxide, SiO2 (silicon carbide, graphite)
- Silicon carbide, SiC (graphite)
Compounds brought to Mars (with optional later in situ production):
- Graphite (for initial steel production)
- Or carbon fiber elements (for construction without steel)
- Platinum chloride (for electroplating anodes, alternative to steel)
- Perovskite solar panels (3.3 tons)
- Mesoporous titanium dioxide, TiO2 (perovskite solar cells, photon trap)
- Gold (perovskite solar cells, conductor)
- Lead iodide (perovskite solar cells)
- N,N-dimethylformamide (perovskite solar cells, solvent)
- Methylammonium iodide, CH3NH3I (perovskite solar cells, reactant)
- 2-propanol (perovskite solar cells, solvent)
- Hydroiodic acid (perovskite solar cells, reactant)
- Spiro-MeOTAD (perovskite solar cells, reactant)
(Prof. John Pisciotta of the West Chester University contributed to this article. Image: NASA/Wikipedia)
This post is also available in Deutsch.




