
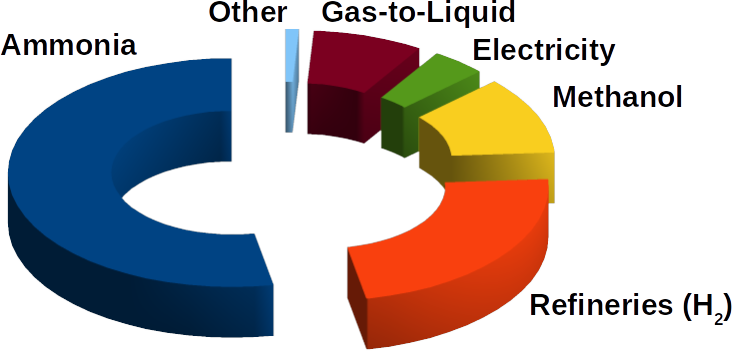
Much research has been done in order to reduce the use of fossil petroleum products as fuels. In that respect syngas (synthetic gas) seems as a great opportunity for sustainable energy developments. Syngas is the mixture composed of hydrogen (H2) and carbon monoxide (CO) as its main components. It represents an important chemical feedstock used widely for industrial processes for generating chemicals and fuels:
Syngas can be produced from methane (CH4) in a reforming reaction with water (H2O), oxygen (O2) or carbon dioxide (CO2). The process called methane dry reforming (MDR) can be combined with carbon dioxide:
CH4 + CO2 → 2 H2 + 2 CO
It is an environmentally friendly path, turning two greenhouse gases into a valuable chemical feedstock.
However, the MDR is process requires chemical catalysts and high temperatures in the range between 700 − 1,000°C. Usually, it suffers from coke deposition and, in consequence, catalyst deactivation.
Some chemists have recently demonstrated that light, and not heat, might be a more effective solution for this energy-hungry reaction.
The photocatalytic solution
A team of researchers at the Rice University in Houston, Texas, together with colleagues from Princeton University and the University of California have developed superior light-stimulated catalysts that can efficiently power MDR reactions without any heat input. This work has been published in the prestigious journal Nature Energy.
They have reported a highly efficient and coke-resistant plasmonic photocatalyst containing precisely one ruthenium (Ru) atom for every 99 copper (Cu) atoms. The isolated single-atom of Ru obtained on Cu antenna nanoparticles provides high catalytic activity for the MDR reaction. On the other side, Cu antennas allow strong light adsorption and under illumination and deliver hot electrons to ruthenium atoms. The researchers suggested that both, hot-carrier generation and single-atom structure are essential for excellent catalytic performance in terms of efficiency and coking resistance.
The optimal Cu-Ru ratio have been investigated in synthesized series of CuxRuy catalysts with varying molar ratios of plasmonic metal (Cu) and catalytic metal (Ru), where x,y are atomic percentage of Cu and Ru. Overall, the Cu19.8Ru0.2 was the most promising composition in terms of selectivity, stability and activity. In comparison to pure Cu nanoparticles, the Cu19.8Ru0.2 mix exhibits increased photocatalytic reaction rates (approx. 5.5 times higher) and improved stability with its performance maintained over 20 h period. Calculations showed that isolated Ru-atoms on Cu lower the activation barrier for the methane dehydrogenation step in comparison to pure Cu without promoting undesired coke formation.
In addition, the research has been supported by different methods (CO-DRIFTS with DFT) in order to unravel and prove single-atom Ru structures on Cu nanoparticles occurring in Cu19.9Ru0.1 and Cu19.8Ru0.2 compositions.
The comparison between thermocatalytic and photocatalytic activity at the same surface for MDR has also been demonstrated. The thermocatalytic reaction rate at 726°C (approx. 60 µmol CH4 / g / s) was less than 25% of photocatalytic reaction rate under white-light illumination with no external heat (approx. 275 µmol CH4 / g / s). This enhancement in the activity is attributed to the hot-carrier generated mechanism which is predominant in the photocatalytic MDR. The role of the hot-carrier is an increase in C−H activation rates on Ru as well as improved H2 desorption.
The scientists also reported the catalyst achieving a turnover frequency of 34 mol H2 / mol Ru / s and photocatalytic stability of 50 h under focused white light illumination (19.2 W / cm2) with no external heat.
As the synthesized photocatalysts is primarily based on Cu which is an abundant element, this approach provides a promising, sustainable catalyst operating at low-temperatures for MDR. This allows cheaper syngas production at higher rates, bringing us closer to a clean burning carbon fuel.
(Photo: Wikipedia)





